Ethoxyethane, C2H5OC2H5, is commonly used as a solvent in the purification of compounds. The boiling point of C2H5OC2H5 is 36 °C. The safety data sheet for C2H5OC2H5 states: ‘Extremely flammable. Keep away from sources of ignition.’ During the purification process, a compound is dissolved in C2H5OC2H5 by heating it for an extended period of time. This is done using glassware that is open to the atmosphere. This step in the purification process should be carried out using a
A. water bath in a fume cupboard.
B. water bath on a laboratory bench.
C. Bunsen burner in a fume cupboard.
D. Bunsen burner on a laboratory bench.
Solution

2) A Chemistry class conducted a practical investigation to determine the calibration factor of a calorimeter using two different methods: electrical and chemical. Each student compared the results from the two different methods and presented the investigation as a scientific poster. The materials, set-up and methods used by the students are shown below (9 marks)
Methods Electrical method for collecting calibration data
1. Add 100 mL of water to the calorimeter. Stir the water and record its temperature every 30 seconds for several minutes.
2. Apply a voltage of 6 V for three minutes. Stir throughout and record the temperature every 30 seconds.
3. Record the voltage and the current while the water is heating.
4. Once the power is turned off, continue to stir the water and record the temperature every 30 seconds for a further three minutes.
Chemical method for collecting calibration data
1. Measure 3.0 g of KNO3 accurately.
2. After completing the electrical calibration, add the KNO3 to the calorimeter.
3. Stir and record the temperature every 30 seconds.
Student A wrote the following aim.
Student A - Aim To compare the calibration factors obtained from two different methods The calibration factors were found by recording the temperature change of a solution resulting from the addition of a measured electrical input and from potassium nitrate dissolving in water.
a. The dependent variable in this investigation is the calibration factor. Identify the independent variable from Student A’s aim.
b. Identify one systematic error that applies only to the electrical method of calibration.
Solution to a) and b)
c. Identify one limitation of the chemical method of calibration, as given above. Explain how it could affect the reliability of the results.

Examine the graphs below prepared by Student A and Student B for the temperature change during electrical calibration.
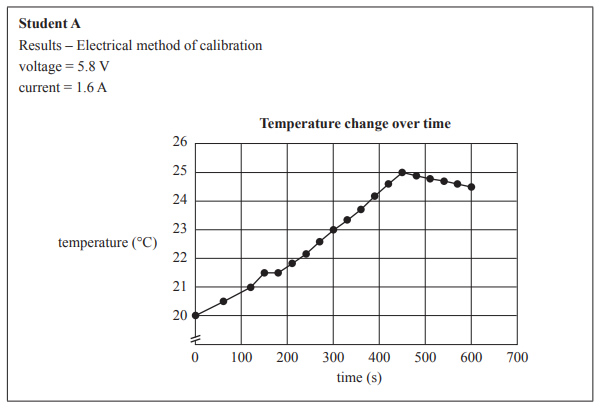
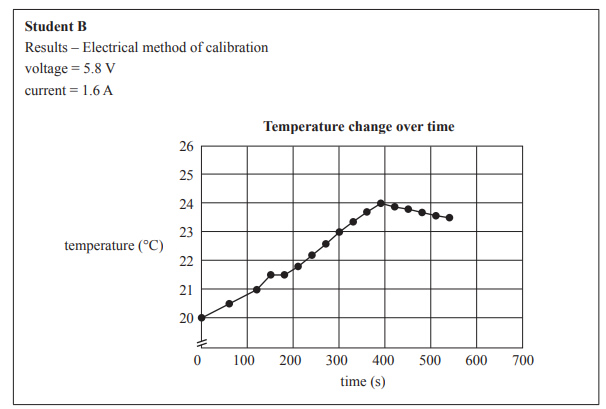
d) Identify one difference in the results between the students’ graphs and suggest what variation in the students’ experiments might account for this difference.
Solution

Student B’s data for the chemical method of calibration is shown in the graph below.
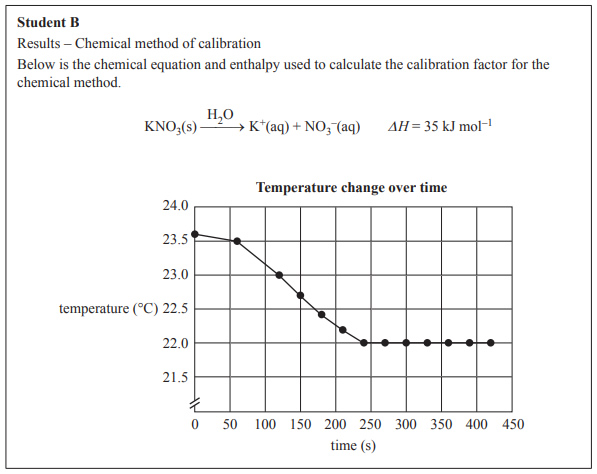
e) Use this data to calculate the calibration factor, in
J °C–1, for the chemical method of calibration.
Solution
